Speciality Products

FROM CONCEPT TO REALITY
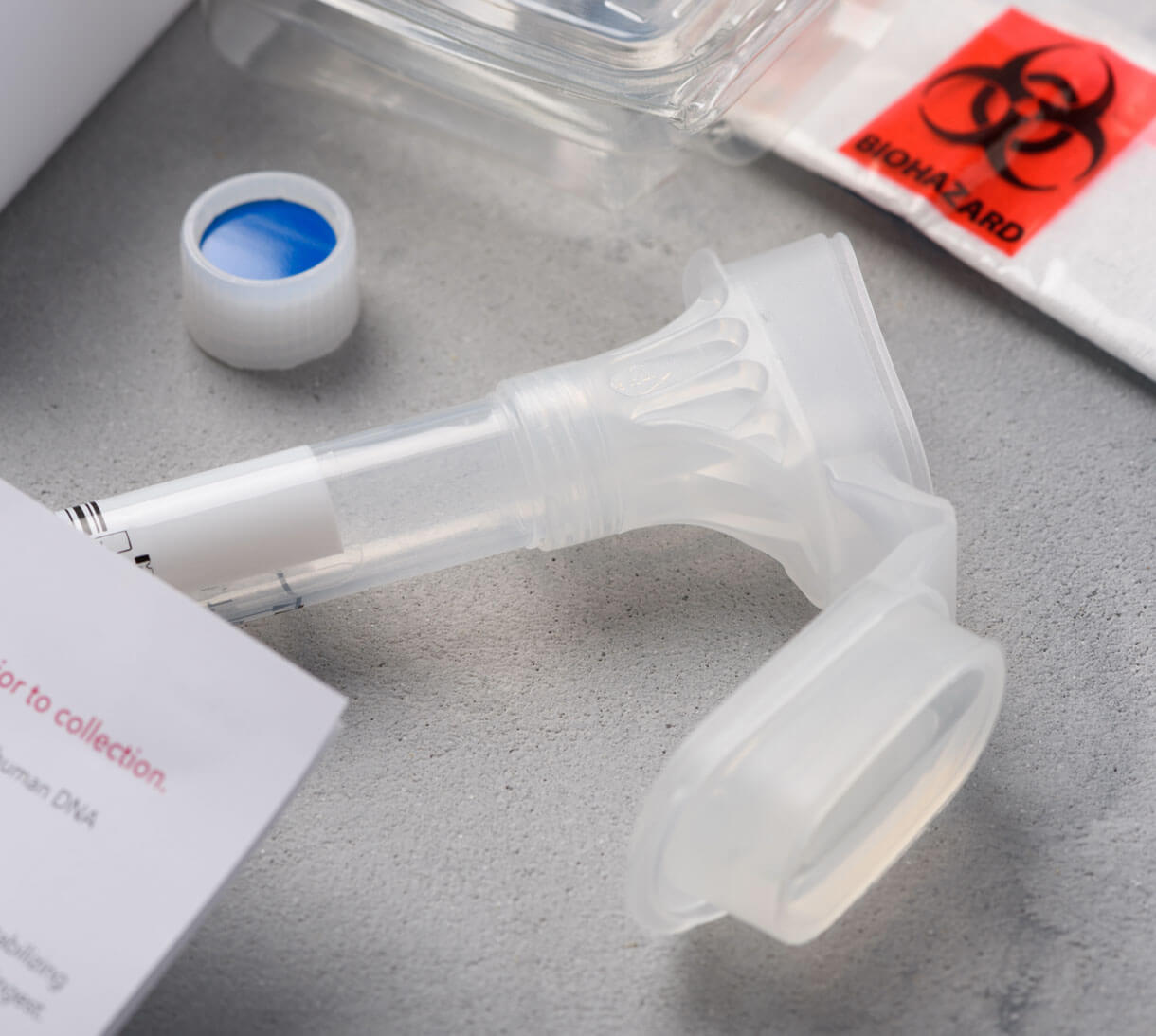
When you have an idea for a new product, you need a Contract Development and Manufacturing Organization (CDMO) to bring it to market. Cenmed can be your CDMO.
With decades of experience in the pharmaceutical and medical device space, Cenmed can take your idea from concept to reality. We are an integrated, nimble, and solutions-driven supplier and manufacturer of custom products.
Custom Manufacturing to
Achieve Your Vision
When you contact us, we'll ask some preliminary questions about your device or formulation, including your IP, design, materials, volumes, and packaging. We'll then discuss our all-in-one approach to design and manufacturing.

-
 How to Get Started
How to Get StartedShare some details about your idea, and we'll assess feasibility and create a proposal.
-
 FDA COMPLIANCE
FDA COMPLIANCEProducts are manufactured in cGMP-compliant facilities.
-
 Fill for Patent
Fill for PatentWe recommend filing a patent application before telling us about your idea. If you have a published patent, provide the patent number.
-
 Your Idea, Our Expertise
Your Idea, Our ExpertiseOur experts are here to help you bring your idea to life.
OUR CAPABILITIES
-

PRODUCT DESIGN
-

3D PRODUCT RENDERING
-

MOLD DEVELOPMENT
-

ULTRASONIC & RF WELDING
-

HOLE PUNCHING
-

AUTOMATED PACKAGING
-

LABELING & PRINTING
-

Assembly Class I & II Medical Devices
-
Cloud Servers & Portals
LET’S TALK ABOUT WHAT YOU'RE LOOKING FOR
Our experts are here to help you bring your idea to life.
